> Seminar & Notice > Seminar
> Seminar & Notice > Seminar
| 제목 | Emerging New Ingredients for Cardiovascular Health (1) | ||||||
|---|---|---|---|---|---|---|---|
| 작성자 | 관리자 | 조회수 | 3765 | ||||
| 등록일 | 2004년 11월 22일 | ||||||
| 첨부파일 | |||||||
|
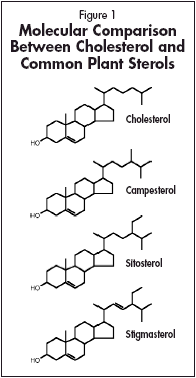
Phytosterols
For over 50 years, plant sterols (also known as phytosterols) and their esters have been studied for a number of reasons including their effects on blood cholesterol in humans. Extensive clinical data in animals and humans indicate that these naturally occurring lipid-soluble compounds can play important roles in the maintenance of healthy cholesterol levels. Plant sterols and stanols can be found in vegetables, fruits, legumes, grains and oils, with some of the most common plant sterols being campesterol, sitosterol and stigmasterol (Fig. 1). As can be seen from their structure, these compounds are chemically similar to cholesterol; however, slight structural differences such as an ethyl group (sitosterol) or methyl group (campesterol) in the side chain differentiate them from cholesterol. Since plant sterols cannot be manufactured in the body, they must be obtained from the diet. Unfortunately, the typical Western diet does not supply enough sterols or stanols to result in significant alterations in blood lipids. It is well understood that cholesterol plays numerous critical roles in the body, from hormone synthesis to helping maintain proper membrane function. Cholesterol can either be derived from the diet or can be synthesized endogenously in the liver. Dietary cholesterol interacts with bile acids and lipases and is ultimately absorbed across the intestinal mucosa where it is packaged into chylomicrons whereupon it makes its way in to the lymphatic system, the liver and ultimately to the systemic circulation. As part of its many functions, the liver also secretes cholesterol into the intestinal tract where it can either be reabsorbed or excreted from the body. One mechanism by which plant sterols appear to function is by competing with cholesterol at the level of the enterocyte. Since the molecular structure of phytosterols is similar to cholesterol, phytosterols can compete with cholesterol for absorption from the gastrointestinal tract. Specifically, plant sterols can displace cholesterol from intestinal micelles, thus inhibiting the absorption of dietary and biliary cholesterol in the gut. Dietary cholesterol, much of which is esterified, is first hydrolyzed in the gastrointestinal tract. The resulting free cholesterol can then be absorbed through mixed micelles, which are combinations of free cholesterol, mono and diacylglycerols, fatty acids, phospholipids and bile salts. This absorption through mixed micelles has recently thought to be due to existing cholesterol transporters in the intestinal mucosa, though earlier evidence indicated it may be due to passive diffusion. Nonetheless, plant sterols, which are more hydrophobic than cholesterol, may displace cholesterol from the mixed micelles, leading to a reduction of micellar cholesterol absorption. A second mechanism by which plant sterols may function to lower cholesterol is by altering the ability of acyl-coenzyme A cholesterol acyltransferase (ACAT) to esterify cholesterol in the enterocyte. A portion of the free cholesterol that has been absorbed in to the enterocyte must be re-esterified prior to being packaged in to chylomicrons. This re-esterification is performed by the ACAT enzyme. If this re-esterification is inhibited, ultimately, the amount of LDL produced by the liver may be reduced.
|
|||||||